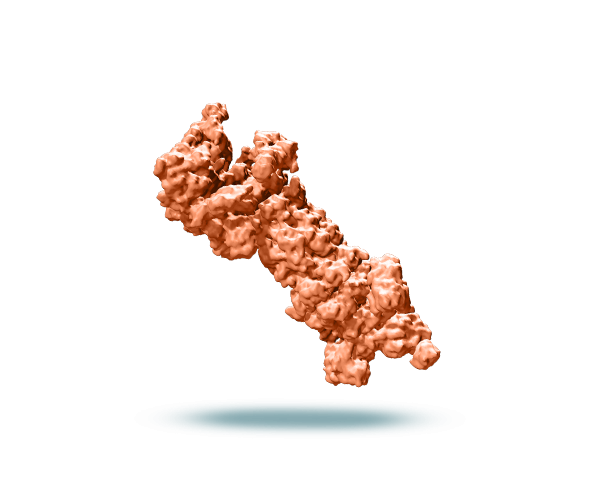
Proteasome
The ubiquitin proteasome pathway is the main pathway for intracellular protein degradation and is essential for normal cell function.1 More than 80% of cellular proteins are degraded through this pathway, including those involved in progression, apoptosis, DNA repair, and protein quality control.2 Proteasomal degradation also activates NF-κB signaling, which upregulates anti-apoptotic factors, cell adhesion molecules, cytokines, and growth factors that promote survival of myeloma cells.3
Due to excess production of immunoglobulins and high rates of protein synthesis, myeloma cells are particularly dependent on the proteasome to survive.1,4 Inhibiting proteasomes in myeloma cells causes accumulation of misfolded and regulatory proteins, which leads to apoptosis.4 Proteasome inhibition has also been shown to trigger cell stress and disrupt the tumor microenvironment.3
Learn more about modalities targeting proteasomes:
small molecules.
Search our clinical trials.
Visit our resources section for additional information on potential oncogenic targets.
NF-kB: nuclear factor kappa-light-chain-enhancer of activated B cells.
1. Moreau P, Richardson PG, Cavo M, et al. Blood. 2012;120(5):947-959. 2. Kubiczkova L, Pour L, Sedlarikova L, Hajek R, Sevcikova S. J Cell Mol Med. 2014;18:947-961. 3. Palumbo A, Anderson K. N Engl J Med. 2011;364(11):1046-1060. 4. Hideshima T, Richardson PG, Anderson KC. Mol Cancer Ther. 2011;10(11):2034-2042.